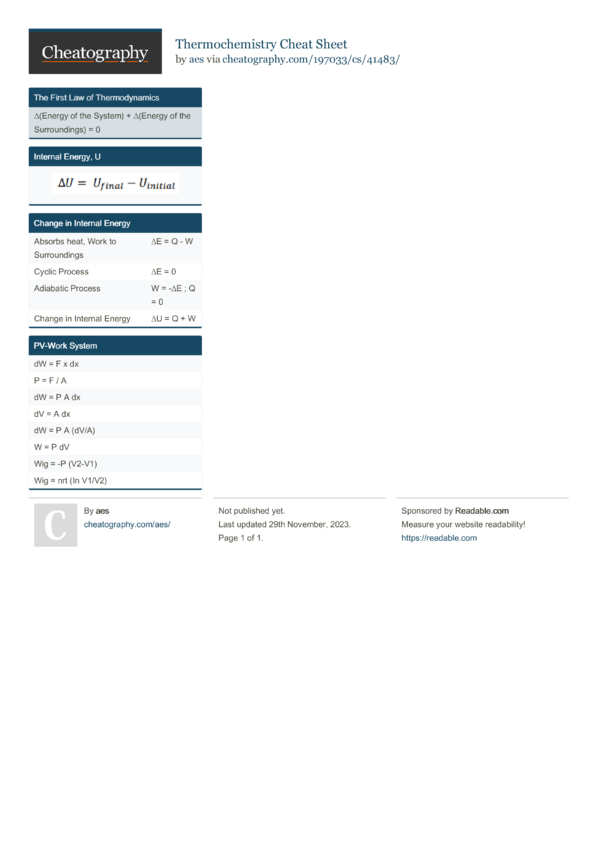
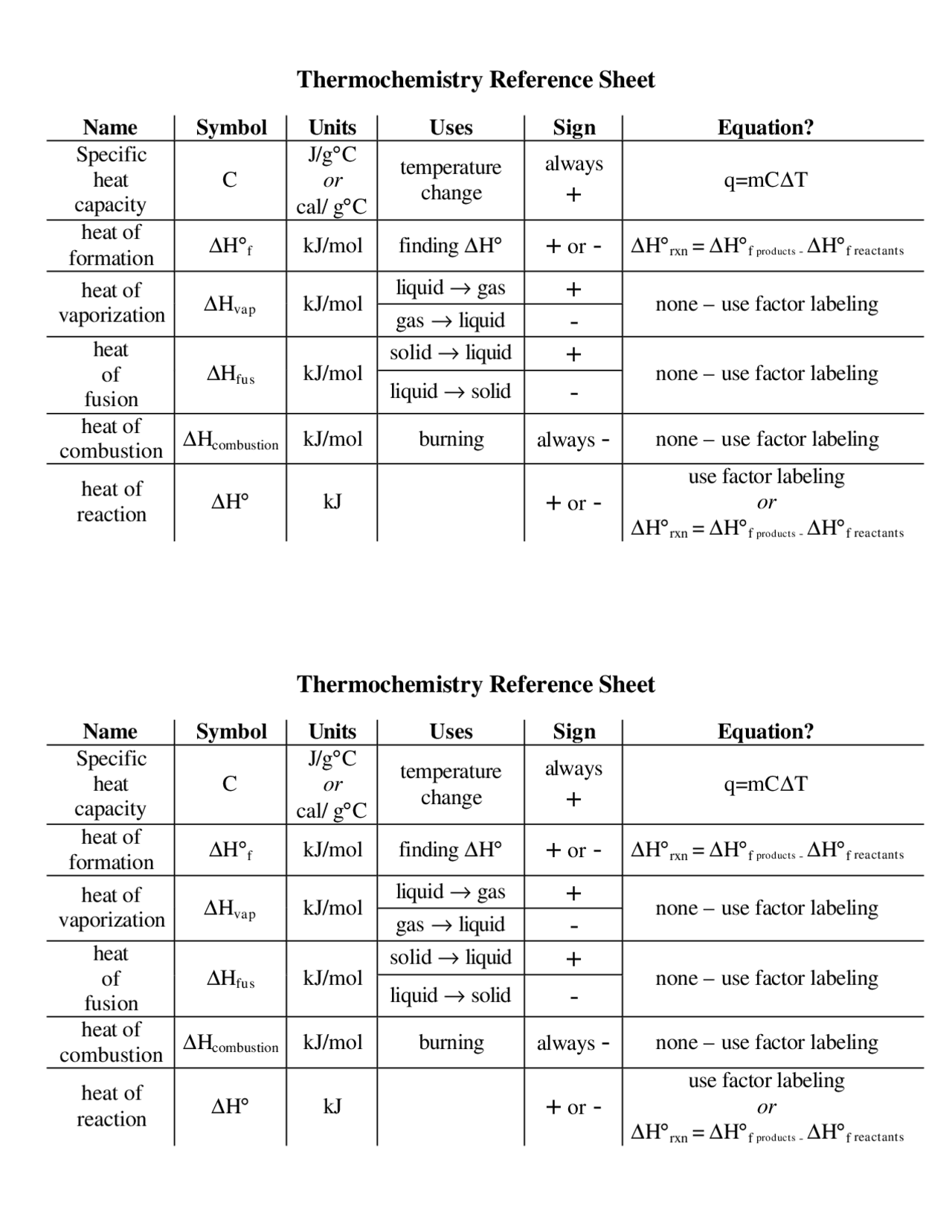
Thermochemistry Cheat Sheet - Does the beaker get warmer or colder? ∂w=−pext⋅∂v and ∂q is the heat transaction between the. Answer the following questions regarding the addition of 14.0 g of koh to water: In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Thermo cheat sheet all systems are closed ( n=0 mol) in general: Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass.
Thermo cheat sheet all systems are closed ( n=0 mol) in general: Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass. In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Answer the following questions regarding the addition of 14.0 g of koh to water: ∂w=−pext⋅∂v and ∂q is the heat transaction between the. Does the beaker get warmer or colder?
In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Thermo cheat sheet all systems are closed ( n=0 mol) in general: Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Does the beaker get warmer or colder? Answer the following questions regarding the addition of 14.0 g of koh to water: Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass. ∂w=−pext⋅∂v and ∂q is the heat transaction between the.
Thermochemistry Cheat Sheet The figure above illustrates the boiling
Thermo cheat sheet all systems are closed ( n=0 mol) in general: Answer the following questions regarding the addition of 14.0 g of koh to water: In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Does the beaker get warmer or colder? Thermochemistry reference sheet.
Thermochemistry Cheat Sheet by aes Download free from Cheatography
∂w=−pext⋅∂v and ∂q is the heat transaction between the. In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Does the beaker get warmer or colder? Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass..
Thermochemistry Equation Sheet
Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Does the beaker get warmer or colder? Answer the following questions regarding the addition of 14.0 g of koh to water: Thermo cheat sheet all systems are closed ( n=0 mol) in general: Thermochemistry reference sheet q = mc∆t variable meaning common units q.
Thermochemistry Equation Sheet
Thermo cheat sheet all systems are closed ( n=0 mol) in general: Does the beaker get warmer or colder? Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass. In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat.
Thermochemistry shortcuts Chemistry education, Teaching chemistry
Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Answer the following questions regarding the addition of 14.0 g of koh to water: ∂w=−pext⋅∂v and ∂q is the heat transaction between the. Does the beaker get warmer or colder? In this chapter we begin an exploration of thermochemistry, the study of the role.
06 thermochemistry
∂w=−pext⋅∂v and ∂q is the heat transaction between the. Thermo cheat sheet all systems are closed ( n=0 mol) in general: Answer the following questions regarding the addition of 14.0 g of koh to water: Does the beaker get warmer or colder? In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the.
THERMOCHEMISTRY PDF
In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Answer the following questions regarding the addition of 14.0 g of koh to water: Thermo cheat sheet all systems are.
Thermochemistry Cheat Sheet Pdf
Thermo cheat sheet all systems are closed ( n=0 mol) in general: Answer the following questions regarding the addition of 14.0 g of koh to water: In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Does the beaker get warmer or colder? Thermochemistry reference sheet.
Chemical thermodynamics Cheat Sheet Chemistry Docsity
Answer the following questions regarding the addition of 14.0 g of koh to water: In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass. Does the.
Thermochemistry formula sheet Cheat Sheet Chemistry Docsity
Answer the following questions regarding the addition of 14.0 g of koh to water: Does the beaker get warmer or colder? ∂w=−pext⋅∂v and ∂q is the heat transaction between the. Learn the basics of thermochemistry, such as endothermic and exothermic reactions, enthalpy, bond energies, and calorimetry. Thermo cheat sheet all systems are closed ( n=0 mol) in general:
Learn The Basics Of Thermochemistry, Such As Endothermic And Exothermic Reactions, Enthalpy, Bond Energies, And Calorimetry.
Does the beaker get warmer or colder? Thermo cheat sheet all systems are closed ( n=0 mol) in general: In this chapter we begin an exploration of thermochemistry, the study of the role that energy in the form of heat plays in chemical. Thermochemistry reference sheet q = mc∆t variable meaning common units q energy being transferred joules, kj, calorie, calorie m mass.
Answer The Following Questions Regarding The Addition Of 14.0 G Of Koh To Water:
∂w=−pext⋅∂v and ∂q is the heat transaction between the.